2 and [Ni(D2O)6](ClO4)2 at Various Temperatures | Semantic Scholar Figure 2 from FIR Spectra of [Ni(H2O)6](ClO4)2 and [Ni(D2O)6](ClO4)2 at Various Temperatures | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ecaf6b0fee69dc0a5060cdb8d7e5d0c7d395a06e/3-Figure2-1.png)
Figure 2 from FIR Spectra of [Ni(H2O)6](ClO4)2 and [Ni(D2O)6](ClO4)2 at Various Temperatures | Semantic Scholar

Nickel transition metal Chemistry nickel(II) Ni2+ complex ions ligand substitution redox chemical reactions principal oxidation states +2 +3 GCE AS A2 IB A level inorganic chemistry revision notes
![Draw the CFT diagram for [Ni(H2O)6]2+ (NO LINKS STRICTLY)!!! - Chemistry - Coordination Compounds - 11664449 | Meritnation.com Draw the CFT diagram for [Ni(H2O)6]2+ (NO LINKS STRICTLY)!!! - Chemistry - Coordination Compounds - 11664449 | Meritnation.com](https://s3mn.mnimgs.com/img/shared/content_ck_images/ck_599e6db7e641a.png)
Draw the CFT diagram for [Ni(H2O)6]2+ (NO LINKS STRICTLY)!!! - Chemistry - Coordination Compounds - 11664449 | Meritnation.com
✓ Solved: Describe the distribution of d electrons in [Ni(H2O)6]^2+, using crystal field theory. How...
![Ni(H2O)6] 2+ (aq) is green in colour whereas [Ni(H2O)4 (en)]2+(aq)is blue in colour, give reason - YouTube Ni(H2O)6] 2+ (aq) is green in colour whereas [Ni(H2O)4 (en)]2+(aq)is blue in colour, give reason - YouTube](https://i.ytimg.com/vi/pONeuFEpHnU/maxresdefault.jpg)
Ni(H2O)6] 2+ (aq) is green in colour whereas [Ni(H2O)4 (en)]2+(aq)is blue in colour, give reason - YouTube
What is the hybridization, transition, spin, colour, magnetism, and geometry of [Ni(H2O) 4] +2? - Quora
2·2Н2О and the complexes M(HTBA)2(H2O)2 (M = Ni, Co, Fe) - ScienceDirect Crystal structure and properties of the precursor [Ni(H2O)6](HTBA)2·2Н2О and the complexes M(HTBA)2(H2O)2 (M = Ni, Co, Fe) - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0277538713008310-fx1.jpg)
Crystal structure and properties of the precursor [Ni(H2O)6](HTBA)2·2Н2О and the complexes M(HTBA)2(H2O)2 (M = Ni, Co, Fe) - ScienceDirect
![Triplet Electronic States in d2 and d8 Complexes Probed by Absorption Spectroscopy: A CASSCF/CASPT2 Analysis of [V(H2O)6]3+ and [Ni(H2O)6]2+ | Inorganic Chemistry Triplet Electronic States in d2 and d8 Complexes Probed by Absorption Spectroscopy: A CASSCF/CASPT2 Analysis of [V(H2O)6]3+ and [Ni(H2O)6]2+ | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/ic0010860/asset/images/ic0010860.social.jpeg_v03)
Triplet Electronic States in d2 and d8 Complexes Probed by Absorption Spectroscopy: A CASSCF/CASPT2 Analysis of [V(H2O)6]3+ and [Ni(H2O)6]2+ | Inorganic Chemistry
![SOLVED: The compound [Ni(H2O)6]Cl2 is paramagnetic. Determine the oxidation number of nickel in this compound, the most likely geometry of the coordination around the nickel, and the possible configurations of the d SOLVED: The compound [Ni(H2O)6]Cl2 is paramagnetic. Determine the oxidation number of nickel in this compound, the most likely geometry of the coordination around the nickel, and the possible configurations of the d](https://cdn.numerade.com/ask_previews/543ac424-a25c-42c6-88c8-ae8f1d1a07d2_large.jpg)
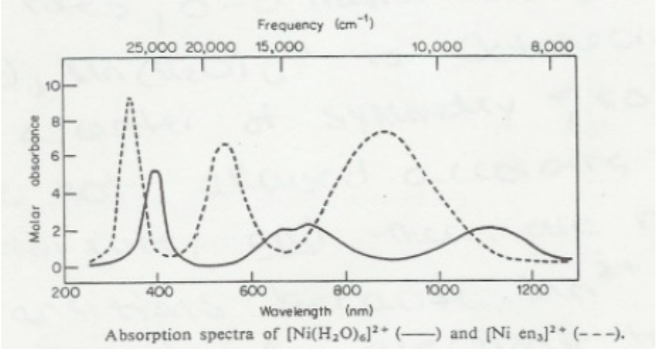




![a) Absorption spectrum of the [Ni(H2O)6]²⁺ complex in the spectral... | Download Scientific Diagram a) Absorption spectrum of the [Ni(H2O)6]²⁺ complex in the spectral... | Download Scientific Diagram](https://www.researchgate.net/profile/Petya-Petkova-7/publication/279288804/figure/fig1/AS:1132476902711296@1647014942438/a-Absorption-spectrum-of-the-NiH2O6-complex-in-the-spectral-region-395-795-nm_Q320.jpg)

![A solution of [Ni(H2O)6]^2 + is green, but a solution of [Ni(CN)4]^2 + is colorless. Why? A solution of [Ni(H2O)6]^2 + is green, but a solution of [Ni(CN)4]^2 + is colorless. Why?](https://haygot.s3.amazonaws.com/questions/1356080_1287162_ans_2cc4810034ce4d16bbf84081945e7afd.jpg)
![Solved 20. When H2O in the [Ni(H2O)6]2+ complex ion is | Chegg.com Solved 20. When H2O in the [Ni(H2O)6]2+ complex ion is | Chegg.com](https://media.cheggcdn.com/media%2F782%2F7824ed9d-14a5-4257-bc8c-05dc5a9cf139%2FphpvCnudZ.png)
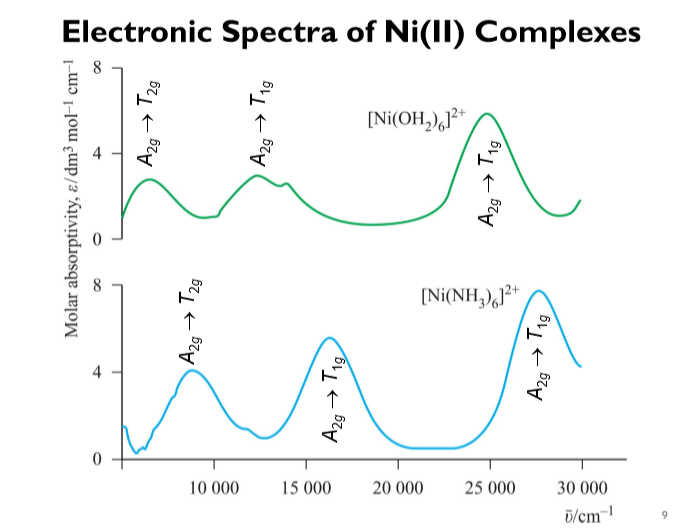
![How [Co(H2O) 6] 2+ is having higher crystal field splitting energy than [Ni( H2O) 6] 2+? - Quora How [Co(H2O) 6] 2+ is having higher crystal field splitting energy than [Ni( H2O) 6] 2+? - Quora](https://qph.cf2.quoracdn.net/main-qimg-fa42f87b83e5b7668101dfb77e274e3c.webp)

![a) Absorption spectrum of the [Ni(H2O)4]²⁺ complex in the spectral... | Download Scientific Diagram a) Absorption spectrum of the [Ni(H2O)4]²⁺ complex in the spectral... | Download Scientific Diagram](https://www.researchgate.net/publication/279288804/figure/fig2/AS:1132476902703108@1647014942540/a-Absorption-spectrum-of-the-NiH2O4-complex-in-the-spectral-region-395-795-nm.jpg)
![A solution of [Ni(H2O)6]^2 + is green but a solution of [Ni(CN)4]^2 - is colourless, Explain. A solution of [Ni(H2O)6]^2 + is green but a solution of [Ni(CN)4]^2 - is colourless, Explain.](https://d1hhj0t1vdqi7c.cloudfront.net/v1/SFk3ZV9DMWxyZ28=/sd/)